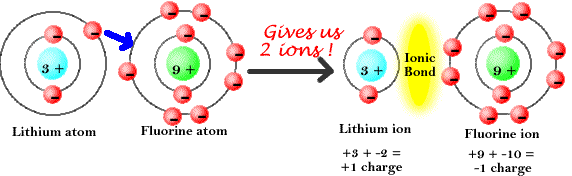
In the previous section of Lesson 1, an atom was described as being a small and dense core of positively charged protons and neutral neutrons surrounded by shells of negatively charged electrons. Conversely, any particle that contains more electrons than protons is said to be negatively charged.Ĭharged Objects as an Imbalance of Protons and Electrons Any particle, whether an atom, molecule or ion, that contains less electrons than protons is said to be positively charged. 
On the other hand, if an atom has an unequal number of protons and electrons, then the atom is electrically charged (and in fact, is then referred to as an ion rather than an atom). Thus, if an atom contains equal numbers of protons and electrons, the atom is described as being electrically neutral. A proton and an electron have an equal amount but an opposite type of charge. The amount of charge on a single proton is equal to the amount of charge possessed by a single electron. The number of electrons that surround the nucleus will determine whether or not an atom is electrically charged or electrically neutral. And an atom containing 8 protons is an oxygen atom. 
An atom containing 6 protons is a carbon atom. An atom containing one proton is a hydrogen atom (H). Atoms of each element are distinguished from each other by the number of protons that are present in their nucleus. There are different types of atoms, known as elements. As discussed in a previous section of Lesson 1, atoms are the building blocks of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
